 
Read more about periodic trends here: brainly. refer to its position on the periodic table and write its valence shell electron configuration. Since Antimony belongs to group 15 and period 5, its valence electrons is 5. Ions is the term for atoms that have gained or lost electrons.Īs said earlier, looking at the periodic table can help us know the number of valence electrons of an atom. That is why it is "electrically neutral" because the atom hasn't gained or lost any electrons. In these groups and periods, elements are together because of their similarities like number of valence electrons, electron affinity, and electronegativity.Įlectrically neutral atoms are atoms that contains no net charge. Elements in the periodic table are arranged in a manner where you can find them in groups and periods. One way to know the number of valence electrons present in an atom is to look at the periodic table and periodic trends. Calcium (Ca) Bromine (Br) Nitrogen (N) Antimony (Sb) Rubidium (Rb) Iron (Fe). 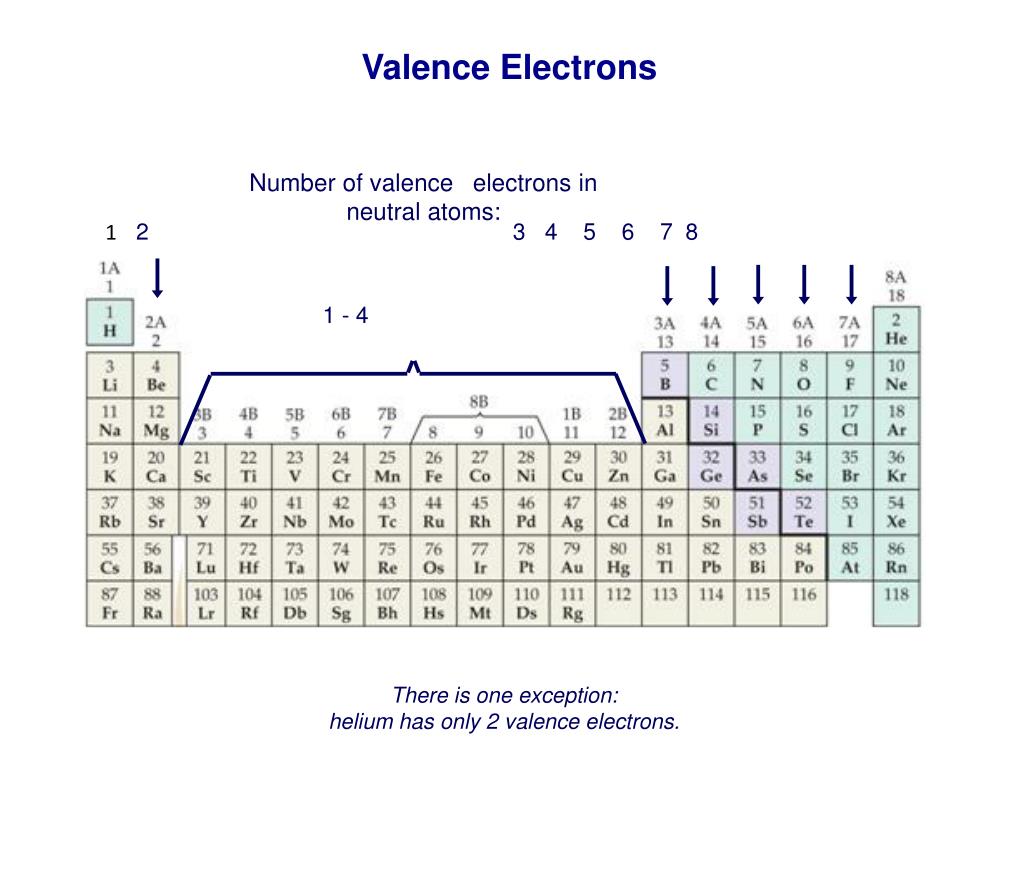
Bond order refers to the number of bonds that can be formed between two atoms. How many electrons are in each of the following shells/subshells. Valence electrons also indicate the bond order of a chemical compound. Antimony pentafluoride SbF5 or F5Sb CID 24557 - structure. They are considered important because they are responsible for some of the element’s chemical properties like the element's electronegativity. Step 2: Find how many more valence electrons are required by one molecule of BrF5: It is. Phosphorus is the eleventh most abundant component, making up. The most common oxidation state of phosphorus is -3. Valence electrons are electrons that lie at the outermost shell of the nucleus' electron orbit. The electron configuration of P is 3s23p3. The number of valence electrons in of an electrically neutral antimony atom is 5. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
